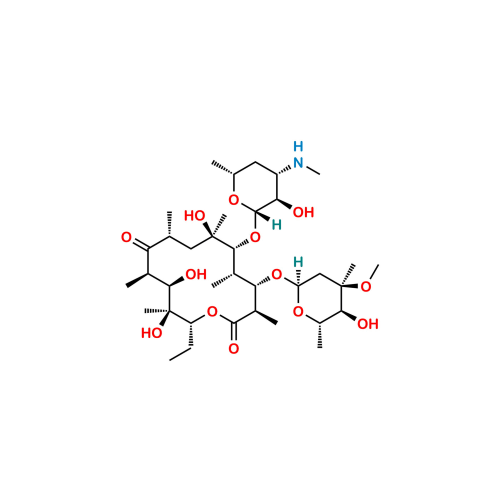
Chemical Name: (3R,4S,5S,6R,7R,9R,11R,12R,13S,14R)-4-[(2,6-dideoxy-3-C-methyl-3-O-methyl-α-L-ribo-hexopyranosyl)oxy]-14-ethyl-7,12,13-trihydroxy-3,5,7,9,11,13-hexamethyl-6-[[3,4,6-trideoxy-3-(methylamino)-β-D-xylo-hexopyranosyl]oxy]oxacyclotetradecane-2,10-dione (as per EP); (3R,4S,5S,6R,7R,9R,11R,12R,13S,14R)-14-Ethyl-7,12,13-trihydroxy-4-{[(2R,4R,5S,6S)-5-hydroxy-4-methoxy-4,6-dimethyltetrahydro-2H-pyran-2-yl]oxy}-6-{[(2S,3R,4S,6R)-3-hydroxy-6-methyl-4-(methylamino)tetrahydro-2H-pyran-2-yl]oxy}-3,5,7,9,11,13-hexamethyloxacyclotetradecane-2,10-dione (as per USP)
Synonym: Erythromycin USP Related Compound N ; 3″-N-Desmethylerythromycin A (EP) ; N-Demethylerythromycin A
Shipping Temperature: Ambient
Country of Origin: India
Smiles: C[C@@H]([C@@H]([C@H](C(O[C@@H]1CC)=O)C)O[C@@](O[C@@H](C)[C@@H]2O)([H])C[C@@]2(C)OC)[C@H]([C@](O)(C[C@H](C([C@@H]([C@@H](O)[C@@]1(O)C)C)=O)C)C)O[C@@](O[C@H](C)C[C@@H]3NC)([H])[C@@H]3O
Erythromycin EP Impurity B is chemically (3R,4S,5S,6R,7R,9R,11R,12R,13S,14R)-4-[(2,6-dideoxy-3-C-methyl-3-O-methyl-α-L-ribo-hexopyranosyl)oxy]-14-ethyl-7,12,13-trihydroxy-3,5,7,9,11,13-hexamethyl-6-[[3,4,6-trideoxy-3-(methylamino)-β-D-xylo-hexopyranosyl]oxy]oxacyclotetradecane-2,10-dione (as per EP); (3R,4S,5S,6R,7R,9R,11R,12R,13S,14R)-14-Ethyl-7,12,13-trihydroxy-4-{[(2R,4R,5S,6S)-5-hydroxy-4-methoxy-4,6-dimethyltetrahydro-2H-pyran-2-yl]oxy}-6-{[(2S,3R,4S,6R)-3-hydroxy-6-methyl-4-(methylamino)tetrahydro-2H-pyran-2-yl]oxy}-3,5,7,9,11,13-hexamethyloxacyclotetradecane-2,10-dione (as per USP). It is also known as Erythromycin USP Related Compound N ; 3″-N-Desmethylerythromycin A (EP) ; N-Demethylerythromycin A. Erythromycin EP Impurity B is supplied with detailed characterization data compliant with regulatory guideline. Erythromycin EP Impurity B can be used for the analytical method development, method validation (AMV), Quality Controlled (QC) application for Abbreviated New Drug Application (ANDA) or during commercial production of Erythromycin.
The product can be used as reference standards and further traceability against pharmacopeial standards (USP or EP) can be provided based on feasibility. Vsquare products are for analytical purpose only and not for human use.